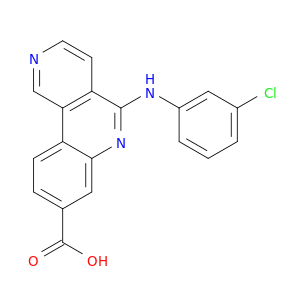
5-[(3-Chlorophenyl)amino]benzo[c]2,6-naphthyridine-8-carboxylic acid
| Title | Journal |
|---|---|
| Identification of potent Yes1 kinase inhibitors using a library screening approach. | Bioorganic & medicinal chemistry letters 20130801 |
| Selectivity, cocrystal structures, and neuroprotective properties of leucettines, a family of protein kinase inhibitors derived from the marine sponge alkaloid leucettamine B. | Journal of medicinal chemistry 20121108 |
| Combined inhibition of EGFR and CK2 augments the attenuation of PI3K-Akt-mTOR signaling and the killing of cancer cells. | Cancer letters 20120901 |
| Identification and validation of inhibitor-responsive kinase substrates using a new paradigm to measure kinase-specific protein phosphorylation index. | Journal of proteome research 20120706 |
| CK2 inhibitor CX-4945 suppresses DNA repair response triggered by DNA-targeted anticancer drugs and augments efficacy: mechanistic rationale for drug combination therapy. | Molecular cancer therapeutics 20120401 |
| Structure-based design of novel potent protein kinase CK2 (CK2) inhibitors with phenyl-azole scaffolds. | Journal of medicinal chemistry 20120322 |
| Synthesis and SAR of inhibitors of protein kinase CK2: novel tricyclic quinoline analogs. | Bioorganic & medicinal chemistry letters 20120101 |
| Antitumoral activity of allosteric inhibitors of protein kinase CK2. | Oncotarget 20111201 |
| 7-(4H-1,2,4-Triazol-3-yl)benzo[c][2,6]naphthyridines: a novel class of Pim kinase inhibitors with potent cell antiproliferative activity. | Bioorganic & medicinal chemistry letters 20111115 |
| Protein kinase CK2 modulates IL-6 expression in inflammatory breast cancer. | Biochemical and biophysical research communications 20111111 |
| Unprecedented selectivity and structural determinants of a new class of protein kinase CK2 inhibitors in clinical trials for the treatment of cancer. | Biochemistry 20111004 |
| Pre-clinical characterization of CX-4945, a potent and selective small molecule inhibitor of CK2 for the treatment of cancer. | Molecular and cellular biochemistry 20111001 |
| Discovery and SAR of 5-(3-chlorophenylamino)benzo[c][2,6]naphthyridine-8-carboxylic acid (CX-4945), the first clinical stage inhibitor of protein kinase CK2 for the treatment of cancer. | Journal of medicinal chemistry 20110127 |
| Structural basis of CX-4945 binding to human protein kinase CK2. | FEBS letters 20110103 |
| Structural determinants of CX-4945 derivatives as protein kinase CK2 inhibitors: a computational study. | International journal of molecular sciences 20110101 |
| CX-4945, an orally bioavailable selective inhibitor of protein kinase CK2, inhibits prosurvival and angiogenic signaling and exhibits antitumor efficacy. | Cancer research 20101215 |